We use sophisticated computer simulation methods to understand the physical origins of biological behavior. Much of our work has focused on membranes and the proteins and peptides that interact with them, but more recently we’ve branched out to work on other topics, including peptide aggregation.
The primary goals of simulation are to interpret existing experiments and suggest new ones. Toward this end, we collaborate with a number of experimental groups.
NOTE: This site will not be maintained much longer. Please update links to point to https://grossfieldlab.org

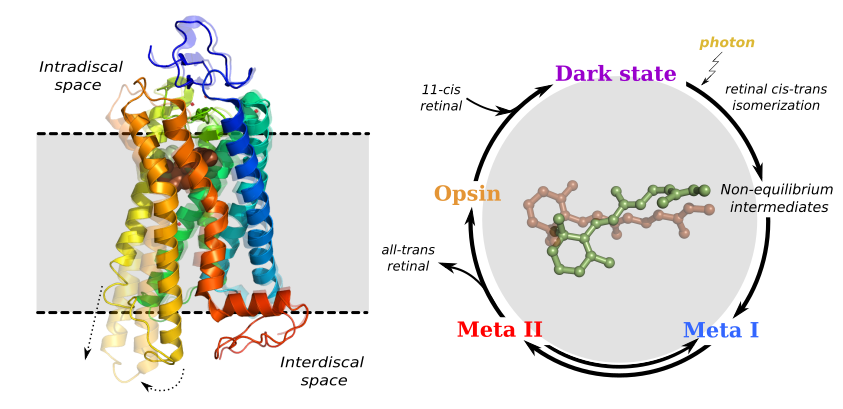
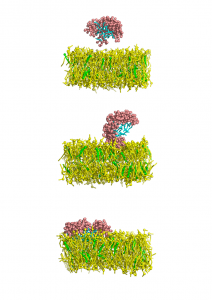 e free energy barrier when approaching neutral membranes.
e free energy barrier when approaching neutral membranes.